| Direct
Methanol Fuel Cell |
| |
| The
Direct Methanol Fuel Cell
is a special case of the
PEM fuel cell. DMFCs and
PEMFCs have similar structures,
two electrodes are separated
by an electrolyte consisting
of a proton-conducting polymer
membrane that is impermeable
to electrons. |
|
|
|
 The
difference between a DMFC and a PEMFC is that
the DMFC uses methanol (CH3OH) as fuel, not
hydrogen. At ambient pressures (1013hPa) methanol
is liquid at temperatures between -97℃ and
64℃. Thus the major advantage of a DMFC is
that its fuel can be handled, stored and transported
similarly to conventional liquid fuels like
gasoline or diesel. On the other hand, methanol
is poisonous and corrosive and DMFCs have
low electrical efficiencies compared to most
other fuel cell types. Applications: electric
motors, portable electricity supply, battery
substitute. The
difference between a DMFC and a PEMFC is that
the DMFC uses methanol (CH3OH) as fuel, not
hydrogen. At ambient pressures (1013hPa) methanol
is liquid at temperatures between -97℃ and
64℃. Thus the major advantage of a DMFC is
that its fuel can be handled, stored and transported
similarly to conventional liquid fuels like
gasoline or diesel. On the other hand, methanol
is poisonous and corrosive and DMFCs have
low electrical efficiencies compared to most
other fuel cell types. Applications: electric
motors, portable electricity supply, battery
substitute.
How a DMFC works
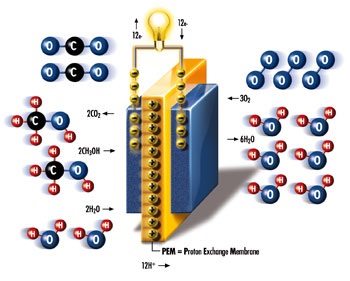
The transparency shows a schematic diagram
of a DMFC. The electrolyte (PEM) is shown
in yellow, the electrodes on either side of
it in blue (anode on the left, cathode on
the right). The anode is supplied with a methanol/water
mixture (red/blue/black and red/blue molecules
on the left). Due to the electrode's catalytic
effect hydrogen is separated from the mixture
and reduced to protons (H+ -ions, yellow +),
yielding free electrons (yellow -) to the
anode. Both protons and electrons now travel
to the cathode side but - very importantly
- on different paths. While the H+- ions pass
through the cell's proton-conducting membrane
the electrons move through the (closed) external
circuit and thereby provide the fuel cell's
electric power (indicated by light bulb).
At the anode the oxygen and carbon left over
from the methanol react with the oxygen from
the water and form carbon dioxide (CO2).
At the cathode the protons that passed through
the membrane and electrons from the external
circuit react with the supplied oxygen (blue)
to form water (red and blue).
Cathode reaction : 3O2
+ 12H+ + 12e- → 6H2O
Anode reaction : 2CH3OH + 2H2O → 2CO2 + 12H+
+ 12e-
Source: h-tec GmbH © h-tec GmbH
|
|
 |
| Direct
Methanol Fuel Cell |
|
